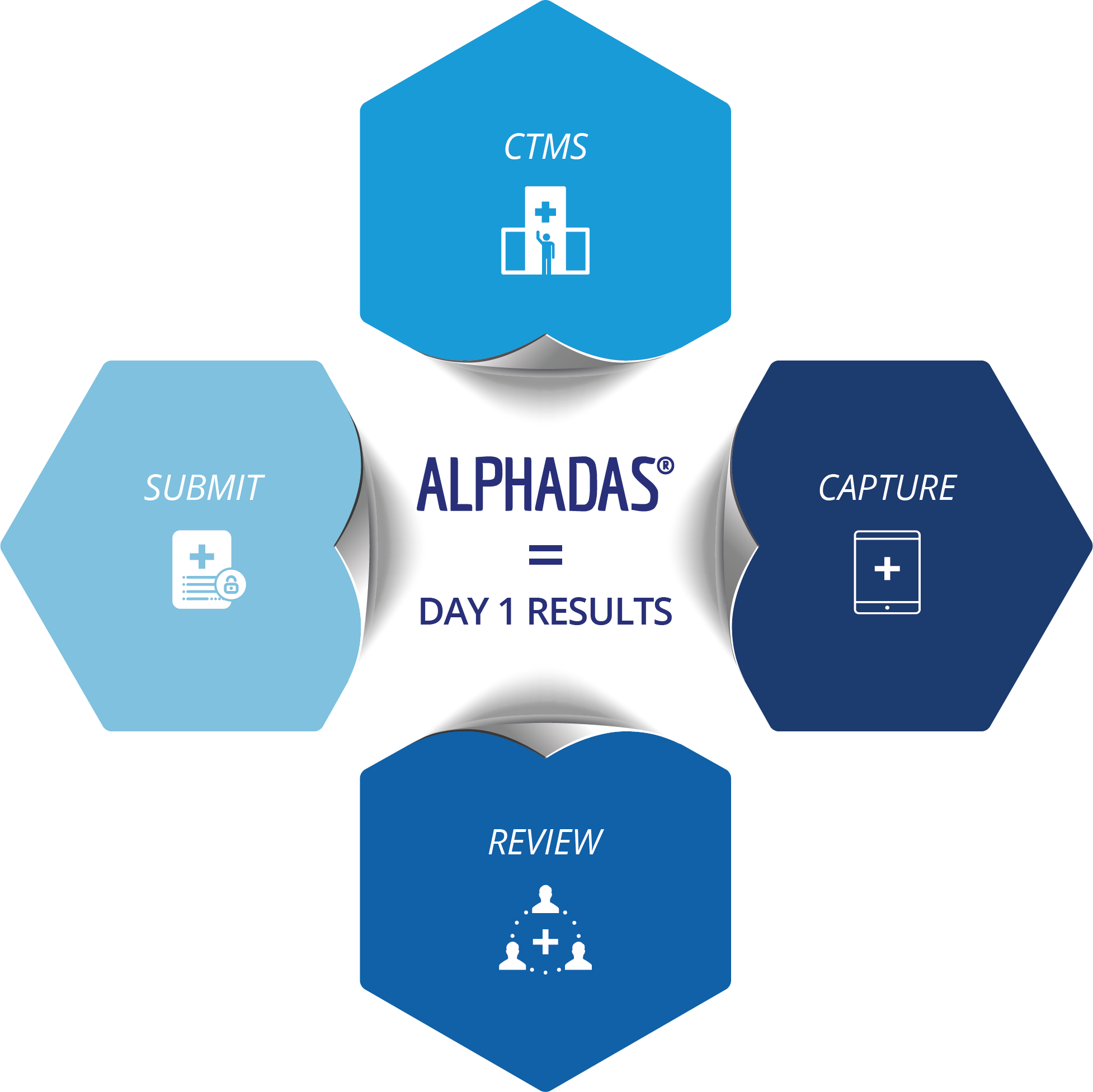
ALPHADAS = Day 1 Results
We believe results are what matter and what makes ALPHADAS and the Instem Clinical team unique. We care about the commitment and investment our clients make in choosing ALPHADAS as their system of choice to streamline their clinical operations and we want them to see a Day 1 Return on Investment. Our early phase expertise is second to none and with more than 100 years of hands-on, practical experience in the Clinical team our clients can be assured that the transformation of how they conduct early phase clinical trials will be handled smoothly and efficiently.
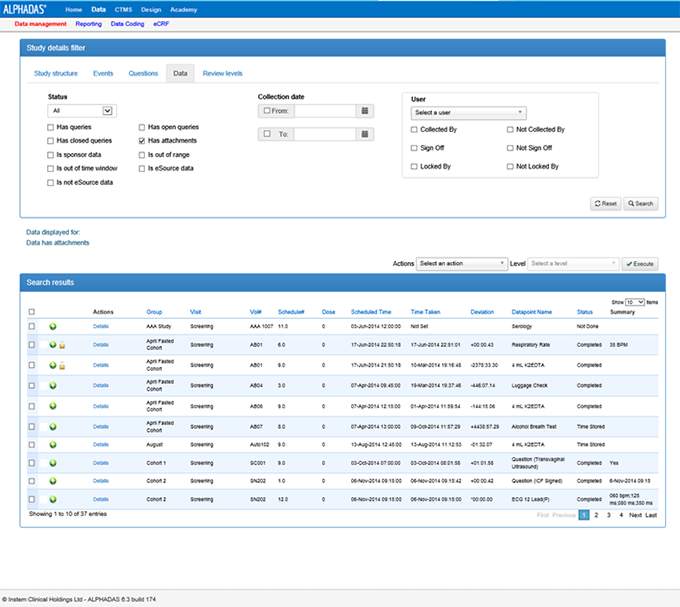
ALPHADAS is a compelling end to end software solution that expedites key drug development decision making and provides operational savings throughout the business. Key benefits include:
- Optimised volunteer/patient identification & recruitment
- Reduced study set up times with flexibility for adaptive study design
- Higher quality data at point of capture
- Comprehensive eCRF/EDC supports multisite data collection & collaboration
- Immediate and accessible data review and exporting
- Direct export of SDTM compliant data
- Full chain of custody from sample collection through to dispatch
- Rapid database lock
- Full offline redundancy
- 21 CFR Part 11 Compliant
Most importantly ALPHADAS provides results on Day 1 of Go Live!
The ALPHADAS Suite
ALPHADAS CTMS
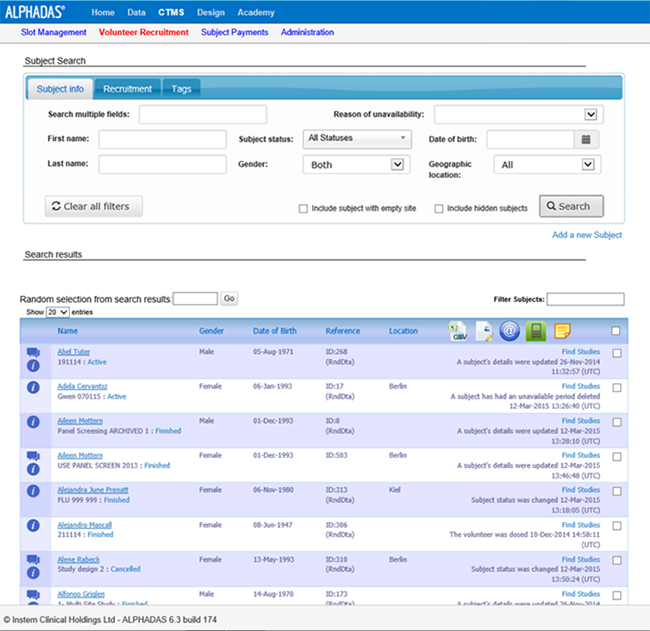
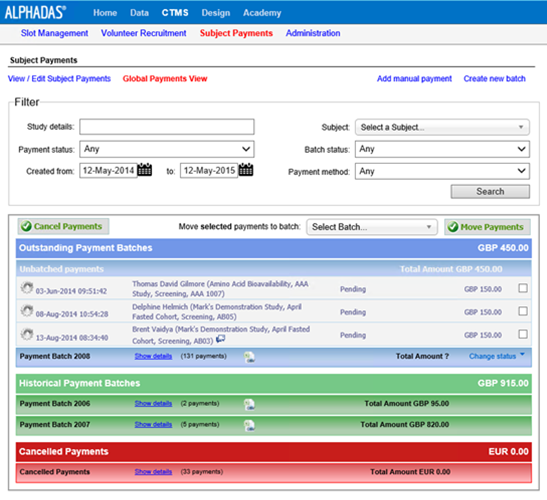
Streamline clinical trial processes from trial start to completion and improve site workflow and efficiencies with tools such as include Volunteer Recruitment and Laboratory Management
ALPHADAS CAPTURE
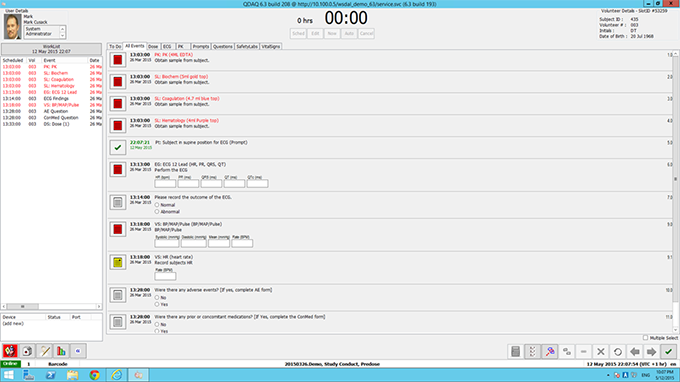
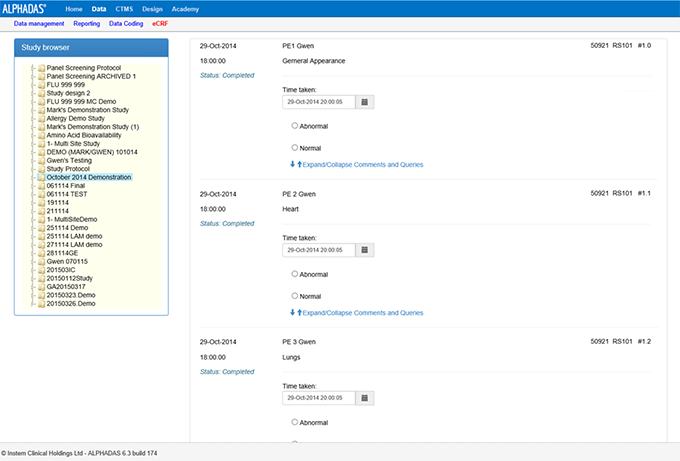
Capture data direct from source instrumentation at bedside so errors are immediately addressed eliminating data queries and enhancing compliance management. Other tools available are our eCRF web based interface and ePRO tablet based patient diary for collection of data by volunteers/patients either onsite or remotely
ALPHADAS REVIEW
Remote real time data monitoring and review, provision of complete management data via the ALPHADAS Dashboard™ and complete audit trail.
ALPHADAS SUBMIT
Regulatory compliant ALPHADAS enables submission of clinical data in a fully SDTM compliant dataset
ALPHADAS can be installed on-site or clients can use Instem’s SaaS (Software-as-a-Service) model to access ALPHADAS offering simpler, more cost effective ways to provide software functionality, maintenance, and support over the Internet. Instem’s professionally managed cloud platforms are run from centralized state-of-the-art data centers, which is being used by clients around the world.
ALPHADAS Academy - Coming Soon
 The ALPHADAS Academy is a sophisticated, easy to use, intuitive web based elearning solution that is available on-demand through the Instem University platform whenever our clients need it. It enhances the service offering we provide and is a value-added extension to our software with the objective of making it easier for clients to use ALPHADAS so that users can better perform their jobs.
The ALPHADAS Academy is a sophisticated, easy to use, intuitive web based elearning solution that is available on-demand through the Instem University platform whenever our clients need it. It enhances the service offering we provide and is a value-added extension to our software with the objective of making it easier for clients to use ALPHADAS so that users can better perform their jobs.
The ALPHADAS Academy meets the needs of all users, from super users through to those staff who may only use ALPHADAS infrequently offering clients even more ways to become proficient on ALPHADAS facilitating increased efficiency and effectiveness and fostering a culture of continuous learning.